Thankyou to the people of The New England
I would like to personally thank everyone who participated in our petition to Save the APVMA.
Yesterday, the 17th of April 2024, the Minister for Agriculture, Murray Watt, flew into Armidale to make the announcement that The Australian Pesticides and Veterinary Medicines Authority (APVMA) will remain in Armidale.
However, of major concern is, The Final Report on the Future Structure and Governance Arrangements for the Australian Pesticides and Veterinary Medicines Authority (APVMA), also known as the Rapid Evaluation, and the Government’s preliminary response has been released.
The recommendation in the report to revoke the government policy order that required APVMA staff to be based in Armidale, has been adopted by the Australian Labor Government, meaning that staff can now be based in Canberra.
This Labor Government, who are vehemently opposed to the decentralisation of government departments and agencies, have taken us for fools. We know, thanks to National Party Senators pressuring the Minister for Agriculture in Senate Estimates, that they have already been taking the jobs away from the New England, revealing that active recruitment for APVMA positions in Canberra has already been occurring over the last 12 months.
Murray Watt and the Labor Government has no vision for Armidale, or regard for our community, blatantly alleging our community is to blame for cultural issues in a government department. If this rule of blaming a location for cultural issues was applied right across the board of departments and agencies, Canberra would be a ghost town!
Thanks to you, for taking a stand for the community of Armidale and the New England, enough pressure was put on them to at least allow part of the Authority operation to remain in Armidale. As we face many challenges of ideological imposition from this cult-like Labor Government in our backyards, it is important that your voices are raised to be heard. Even though the announcement yesterday was not the response we were wanting to hear, at least we have made enough noise to tell the Labor Australian Government that we will not be insulted and punished for having a vision.
If you would like to be kept up to date with issues in the New England Electorate, please subscribe to my e-newsletter here. Thankyou again for your participation in our Petition, and please don’t hesitate to contact my Tamworth or Tenterfield offices on the numbers below or via email at Barnaby.Joyce.MP@aph.gov.au
TIMELINE OF APVMA FIGHT TO STAY IN ARMIDALE
MINISTER FOR AGRICULTURE, FISHERIES AND FORESTRY, SENATOR MURRAY WATT, ANNOUNCES THE APVMA WILL REMAIN IN ARMIDALE, BUT REMOVES CLAUSE TO OFFICIALLY ALLOW CANBERRA RECRUITMENT
17 April 2024
The Final Report on the Future Structure and Governance Arrangements for the Australian Pesticides and Veterinary Medicines Authority (APVMA), also known as the Rapid Evaluation, and the Government’s preliminary response has been released.
The recommendation in the report to revoke the government policy order that required APVMA staff to be based in Armidale, has been adopted by the Australian Labor Government, meaning that staff can now be based in Canberra.
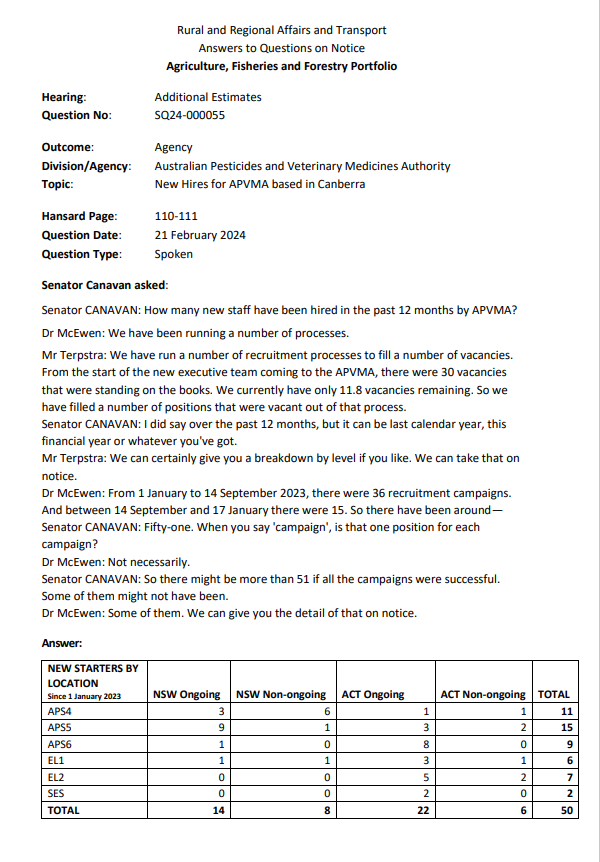
SENATOR CANAVAN ASKS IN THE RURAL AND REGIONAL AFFAIRS AND TRANSPORT SENATE ESTIMATES THE APVMA 'HOW MANY STAFF HAVE BEEN HIRED AND WHERE?'
21 February 2024
MEMBER FOR NEW ENGLAND MAKES REPRESENTATION TO MINISTER FOR AGRICULTURE, FISHERIES AND FORESTRY, SENATOR MURRAY WATT
11 September 2023
Mr Barnaby Joyce made representation to Senator Watt, requesting urgent consideration of the community concerns of the Authority being relocated back to Canberra, reportedly relayed to the APVMA community during a visit from Mr Ken Matthews AO, a former public servant, suggesting this could occur through the slow attrition of staff and that staff should prepare for options.
MINSITER FOR AGRICULTURE, FISHERIES AND FORESTRY, SENATOR MURRAY WATT, ANNOUNCES A REVIEW OF THE OPERATIONS, CONDUCT AND GOVERNANCE OF THE APVMA
14 July 2023
A review commissioned by Minister Watt, undertaken by law firm Clayton Utz was released.The review drew a correlation to the decision made by former Agriculture Minister Barnaby Joyce to relocate the APVMA to Armidale as one of the key factors behind the demise of good governance at the institution. The report also found that the decision to move the regulator to Armidale resulted in a loss of corporate knowledge, a loss of corporate culture and a loss of experience and knowledge of public sector values. If only correlations were considered of any other government department or agency that goes through the machinery of government process, that is where you employ qualified people to do a job, to change culture and invest in developing employees knowledge and skill sets. A well researched indicator of workplace cultural issues has been found to be unscheduled absenteeism. According to the State of the Service Report 2022, delivered by the Australian Public Service Commission there are 61 other Australian government agencies and departments that rank higher than the APVMA with unscheduled absenteeism. The rationale of the selection of agency number 62 of 96 reported must also be questioned. For example, are there reports being commissioned by all Ministers to draw correlations between the physical location, the cultural environment and the departmental or agency corporate culture.
One can only draw their own correlation here that this is purely an attack on the regions, decentralisation and on Armidale.
MINISTER FOR INFRASTRUCTURE, TRANSPORT, REGIONAL DEVELOPMENT AND LOCAL GOVERNMENT
Question No. 58
Mr Barnaby Joyce asked the Minister for Infrastructure, Transport, Regional Development and Local Government, in writing, on 30 November 2022:
In respect of the Agvet Chemical and Technology Innovation Centre of Excellence, CropLife Australia and University of New England, Armidale program and its previously identified funding source (Energy Security and Regional Development Plan):
1. What funding has been identified over the four-year forward estimates as a consequence of the October 2022-23 budget.
2. What is the program or funding source for the project since the change of Government.
3. What is the estimated completion timeframe and any other milestones.
4. What is the expenditure to date for the project.
Ms King - The answer to the honourable Member’s question is as follows:
The former government did not appropriate the March 2022-23 Budget. This means measures and associated election commitments made by the former government such as the Energy Security and Regional Development Plan were not funded

The Hon. Barnaby Joyce MP
Shadow Minister for Veterans’ Affairs
Member for New England
Media Release
30 October 2023
REGIONAL AUSTRALIA AND THE NEW ENGLAND CALLS OUT ALBANESE FOR PUNISHING OUR COMMUNITIES
Across the nation in regional areas, everyday Australians that are tired of watching our nation and our livelihoods being ripped apart are mobilising.
Last week, the National Farmers’ Federation (NFF) was forced to take the extraordinary step of launching its largest protest campaign in almost 40 years, as Labor continues to decimate the industry and the regions.
Member for New England, Barnaby Joyce, said the NFF was desperately trying to save farming from a Labor government that had torn up farming confidence. New NFF President, David Jochinke, also gave a dire warning at the NFF Conference that it was in the fight of its life.
“It has taken just 18 months for Agriculture Minister, Murray Watt, and the Prime Minister to lose the confidence of the agriculture sector,” Mr Joyce said.
“Our farmers are under siege. NFF’s public campaign, ‘Keep Farmers Farming’, is an embarrassment for Minister Watt and the Albanese Labor Government.
“Every single Australian is feeling the impact of the pain and hurt being put onto farmers, because the end result of Labor’s bad policies are families being forced to pay more for food at the checkout, while the producers of the food are receiving less at the paddock gate.
“Farmers are being punished by Labor in almost every possible area, from fresh food tax, scrapping the Ag visa, banning live sheep exports through to the ridiculous and unachievable renewable energy policies that will only hurt agricultural land while power prices go up, reliability goes through the floor, and the profits go overseas.
“Labor’s ludicrous ideology is putting our food and fibre production at risk, while our cost of living goes through the roof. In the electorate of New England, our City of Armidale has been directly targeted by Minister Watt, with the looming announcement of the future of the Australian Pesticides and Veterinary Authority location (APVMA), which could rip out 144 direct jobs from the Armidale community.
“It is heartbreaking and offensive for a Labor government to come along and blame a whole regional community for a workplace issue. Labor do not believe in the same future that we do for our regional city, to build the potential for our university, to build local jobs and most of all in government departments and agencies to be based outside of Canberra. For this reason, I have started a petition to take the voices of the electorate and the agricultural sector to Canberra, to save the APVMA. To sign the petition, visit: https://barnabyjoyce.com.au/save-the-apvma
We can see from the recent referendum just how out of touch the Canberra bubble and the Labor Government are from the rest of Australia. Now, more than ever, we need to decentralise government departments to regional areas, where practical measures, initiatives and policy can be developed within industries and communities that the policies are designed to serve.
Well Minister Watt, if you want to test your performance as a Minister of the Albanese Government within the sector you represent, I think you and Minister Bowen could do the best test of all and that’s a pub test, in the New England. Let’s see how that goes down.
MEDIA RELEASE
11 February 2019
APVMA ON TRACK AND ON SCHEDULE
Federal Member for New England, Barnaby Joyce, has dismissed reports which claim the Australian Pesticides and Veterinary Medicines Authority (APVMA) is behind schedule. “In fact, it’s quite the opposite - the new APVMA building in Armidale is nearing the final stages of completion and there is strong interest in local jobs,” Mr Joyce said. The APVMA received nearly 300 applications for between 15 and 40 jobs during a recent recruitment round. There were 79 applications from scientists all over Australia for 19 science jobs. It is expected the new Armidale premises will house the 150-strong majority of APVMA’s staff by mid this year. What’s more, the latest performance statistics (September 2018 quarter) released in November 2018, show that the APVMA has significantly improved, with 86 per cent of applications finalised within the legislated timeframes. This is the fifth straight quarter of timeframe performance improvement. Even Labor concedes “overall performance figures seem to have improved”. “What this shows is decentralisation is working, bringing more jobs, greater wealth and a better standard of living for people in Armidale and the New England,” Mr Joyce said. (ENDS)

